What is SAS
Synthetic Amorphous Silica explained
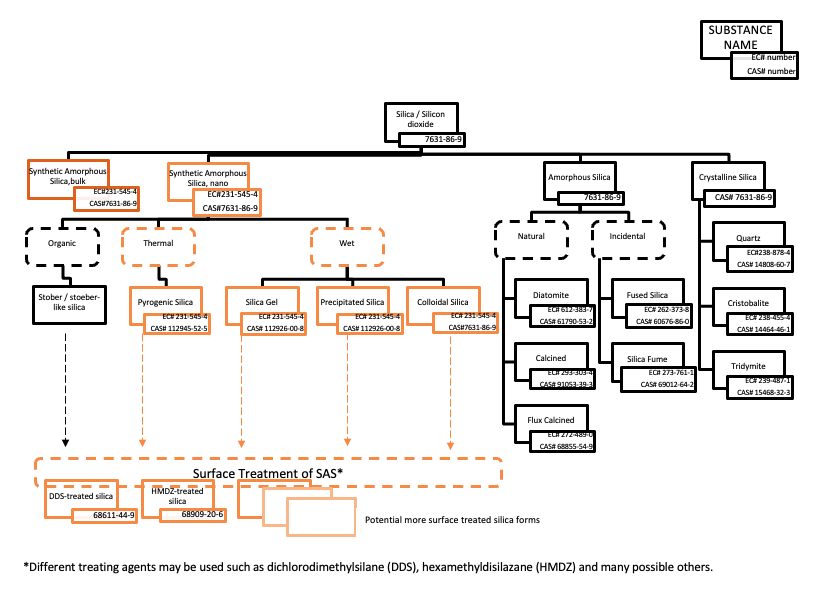
Synthetic Amorphous Silica (SAS) is intentionally manufactured highly pure, crystalline-free, silicon dioxide (SiO2). It has been produced and marketed for almost a century without significant changes in its physicochemical properties and exists in four forms:
- pyrogenic (also called fumed) silica
- precipitated silica
- silica gel
- colloidal silica
Silica also occurs naturally in several forms, the most obvious being sand. It can also be found in other minerals and in many plants such as bamboo, rice and barley. In nature the most abundant form, is crystalline quartz. While some of the silica types have individual Chemical Abstract System (CAS) numbers, the European Community number (EC number) for all silica is 231-545-4.
Silica, independent of its form and method of preparation (including by-products) is found under this number. However, as the forms differ in their hazards to human health, it is essential to distinguish between crystalline silica and Synthetic Amorphous Silica (which is crystalline-free).